Models
Numerical models based on kinetic and mechanistic data derived from laboratory studies can be used to aid our understanding of the composition of the atmosphere, and can be used to simulate concentrations of short-lived free radical species such as OH and HO2. Field measurements of these species therefore provide an ideal test for atmospheric models, and a combination of field measurements and modelling studies can be used to examine our understanding of the atmosphere. Modelling studies provide valuable insight to atmospheric chemistry on local, regional and global scales, and can be used to inform policy decisions regarding pollution, public health and climate change.
By doing such work we hope to provide a link back to laboratory studies to identify areas in which current recommendations for the kinetics and mechanisms used in atmospheric models may require improvement.
Master Chemical Mechanism (MCM)
The Master Chemical Mechanism or MCM is a near-explicit chemical mechanism describing the atmospheric chemistry of primary emitted volatile organic compounds (VOCs). Version 3.2 contains methane and 142 non-methane primary VOCs. The mechanism can be extracted for use in numerical models of atmospheric chemistry and is used in many of the group's modelling studies.
DSMACC
 In collaboration with Prof. Mat Evans (School of Earth and Environment, now at University of York), the Dynamically Simple Model of Atmospheric Chemical Complexity (DSMACC) box model has been used in conjunction with the MCM to investigate the fast photochemical processes responsible for determining the atmospheric concentrations of species such as OH and HO2 in environments ranging from cold Arctic regions to tropical forests and remote tropical islands.
In collaboration with Prof. Mat Evans (School of Earth and Environment, now at University of York), the Dynamically Simple Model of Atmospheric Chemical Complexity (DSMACC) box model has been used in conjunction with the MCM to investigate the fast photochemical processes responsible for determining the atmospheric concentrations of species such as OH and HO2 in environments ranging from cold Arctic regions to tropical forests and remote tropical islands.
GEOS-Chem
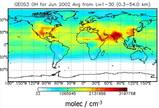 The 3-dimensional global chemistry transport model GEOS-Chem has been used to investigate the impacts of recent laboratory findings on global modelling.
The 3-dimensional global chemistry transport model GEOS-Chem has been used to investigate the impacts of recent laboratory findings on global modelling.
